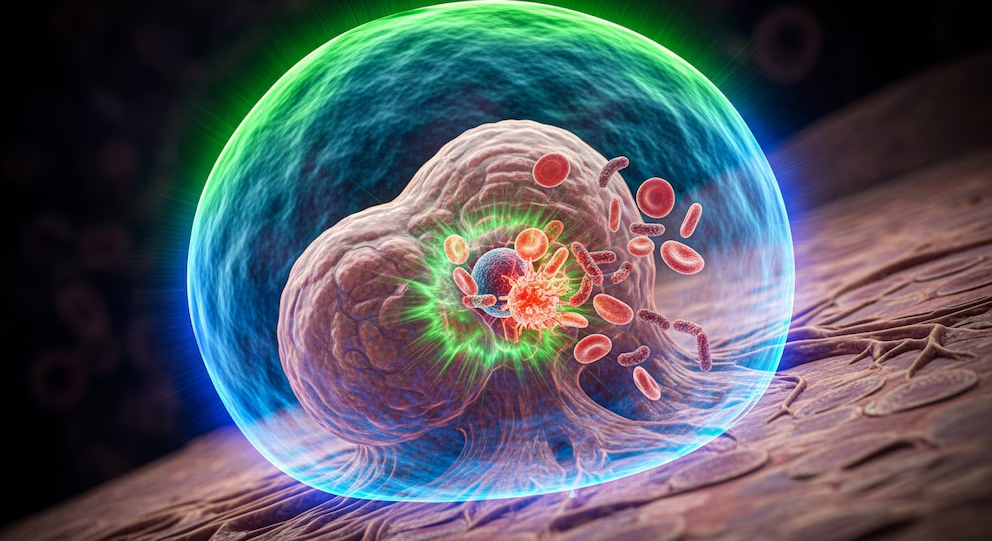
For decades,
treatment resistance was one of the most
complex enigmas in oncology. Why do some tumors stop responding even to advanced therapies? Recent research provides a different answer to the traditional one and proposes that the secret lies not only in genetic mutations, but in the
adaptability of the cancer cells themselves.
The study, developed by scientists from NYU Langone Health and published in the journal Nature, describes a mechanism that allows tumors to modify their behavior reversibly to survive treatments.
This finding introduces a new way of understanding resistance and opens the door to different therapeutic strategies.
Tumor Epigenetic Flexibility
Until now, the dominant theory held that resistance appeared when some cells acquired mutations in their DNA. These infrequent changes gave them advantages against drugs and allowed them to multiply while other cells were eliminated.
The new model does not rule out that process, but adds a key element: cells can also change how they use their genes without altering their structure. This phenomenon is known as epigenetic regulation.
NYU Langone Health study reveals that epigenetic regulation allows tumors to survive drugs without permanent mutations.
We recommend reading:HPV vaccine: why is it so important to prevent cervical cancer?
To understand it, think of DNA as an instruction manual. Epigenetics doesn't modify that manual, but it does decide which instructions are activated and which are paused. In this way, the same cell can behave in different ways depending on the context.
This flexibility allows for rapid responses to adverse situations, such as exposure to a medication. Instead of waiting for a mutation, the cells reorganize their functioning almost immediately.
The Central Role of AP-1 Proteins
The study identifies a group of p
roteins called AP-1 as protagonists of this process. These molecules act as
“switches” that regulate the activity of hundreds of genes.
A distinctive feature of AP-1s is their ability to combine with each other. They form pairs —known as
dimers— and each combination activates a different set of genes. This generates a great diversity of possible responses within the same cell.
As explained by researcher
Gustavo S. França, this system works as an
“evolutionary algorithm”. In practice, the cells test different configurations: those that favor their survival are maintained, while the less effective ones are discarded. Scientists propose anti-adaptation agents to block the adjustment capacity of tumors and improve the response to oncological therapies (Illustrative Image Infobae)
This mechanism of
trial and error allows the tumor to quickly find ways to resist treatments. One of the most relevant findings is that these adaptations are not lost. Through what is called
epigenetic memory, cells transmit these configurations to their descendants.
This means that, over time, cell populations become increasingly resistant. Even when more sophisticated therapies are applied, the tumor already has a repertoire of responses that allows it to survive.
The model was led by
Itai Yanai, who points out that this progressive adaptation capacity helps explain why some advanced cancers are so difficult to treat.
A Dynamic System of Molecular Survival
Researchers describe this system as a kind of
"survival kit" internal. Thanks to the AP-1 proteins, cells can explore multiple combinations of genetic activity without needing to permanently modify their DNA.
This approach changes the way of thinking about cancer evolution. It is no longer just about accumulated mutations, but also about dynamic adjustments that allow cells to adapt to the environment. In addition, these modifications are reversible. This implies that, in theory, they could be blocked or modified with new therapies.
New strategies to confront resistant tumors
The combination of traditional and anti-adaptive therapies is emerging as a promising strategy to combat tumor resistance (Illustrative Image Infobae)
Based on these findings, scientists propose a change in the therapeutic strategy. Instead of focusing solely on eliminating cancer cells, they suggest
intervening on their ability to adapt.
One of the most promising lines is the development of so-called "anti-adaptation agents." These are drugs designed to prevent cells from adjusting their behavior in response to treatments.
According to the NYU Langone Health team, combining these therapies with traditional treatments could increase their effectiveness and prolong the response, making it difficult for the tumor to develop resistance.
This approach essentially seeks to block the "learning" of malignant cells, preventing them from finding new ways to survive.
Mechanisms Shared with Normal Physiological Processes
The study also highlights that this system is not exclusive to cancer. AP-1 proteins participate in normal bodily functions, such as memory formation and tissue repair. This suggests that it is a fundamental biological mechanism, which in the case of tumors is exploited in an uncontrolled manner. The use of single-cell analysis and CRISPR gene editing will allow the identification of AP-1 combinations associated with different types of tumor resistance (Illustrative Image Infobae)
Understanding how it works in normal and pathological conditions will be key to developing therapies that act with precision without affecting essential processes.
The goal now is to decipher in detail
how the different AP-1 combinations operate. To achieve this, scientists plan to use advanced tools such as
single-cell analysis — which studies individual cells — and
genetic editing using CRISPR.
These techniques will allow identifying which configurations are associated with different types of resistance and designing more specific treatments. The challenge is to map this internal "language" of the cells to anticipate their responses and act before they develop defense mechanisms.
This model introduces a broader perspective on tumor resistance. By incorporating the ability to adapt as a central factor, it offers a more complete explanation of why some treatments lose effectiveness.
More importantly, it opens up new possibilities for the development of more durable therapies. Instead of solely pursuing mutations, science is beginning to focus on the dynamic behavior of cells.